Introduction: Anti-CD19 chimeric antigen receptor T cell (CD19CAR-T) therapy has achieved significant clinical outcomes and changed treatment strategy in B-cell malignancies. However, recurrence after CAR-T therapy remains a major obstacle due to poor peak expansion and short persistence of CAR-T cells. We have reported that CD19CAR-T incorporating a co-stimulatory domain consisting of CD79A and CD40 (79A40CAR) provided superior anti-tumor efficacy and survival compared to CD19CAR-T with CD28 or 4-1BB (CD28CAR/4-1BBCAR) in mice xenograft models. 79A40CAR exhibited sustained NF-κB phosphorylation and superior in vivo efficacy compared to the CD28CAR or 4-1BBCAR. (Molecular Therapy 2021, Julamanee J et al.) In this study, we aimed to clarify the molecular mechanism of 79A40CAR by comparing with CD28/4-1BBCAR in terms of T-cell metabolism.
Methods: CD8+T cells were isolated from PBMCs of healthy donors and retrovirally transduced with either CD28IC-, 4-1BBIC-, or 79A40IC-CD19CARs. CD28CAR, 4-1BBCAR, or 79A40CAR were cocultured with 100 Gy irradiated NALM6 at E:T ratio = 1:5, respectively. Repeated antigen stimulations (Ag-stim) were performed, and day 7 after each stimulation was subjected to analysis. RNA-seq, expansion capacity, T-cell phenotype, and metabolic evaluation using a seahorse flux analyzer (Agilent Technologies) were performed. After Ag-stim, CD45RA negative CD62L positive and CD45RA negative CD62L positive CAR-T cell fractions were sorted as central memory (T CM) and effector memory (T EM), respectively. T CM and T EM received Ag-stim. After Ag-stim, we performed evaluation of mitochondrial functionality with MitoTracker based on mitochondrial mass and membrane potential.
Results: All CD19CAR genes were successfully transduced into primary CD8+ T-cells. To understand the differences between co-stimulatory domains, we first confirmed bulk RNA-seq, expansion capacity, and T-cell phenotype. After Ag-stim, 79A40CAR showed enhancement of the NF-κB pathway. We detected few DEGs when 79A40CAR compared to 4-1BBCAR, however, we detected 162 upregulated DEGs when 79A40CAR compared to CD28CAR. Principal component analysis of gene expression demonstrated similarity between 4-1BBCAR and 79A40CAR. We confirmed that 79A40CAR had superior expansion capacity than CD28, or 4-1BBCAR after repeated Ag-stim. PD-1 and Tim-3 expression in 79A40CAR was lowest after repeated Ag-stim. In the evaluation of T-cell differentiation, 4-1BB and 79A40CAR showed an earlier transition to the effector phenotype compared to CD28CAR after Ag-stim.
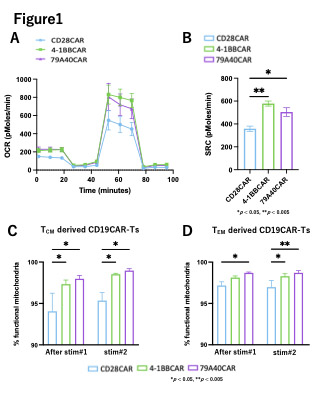
To evaluate differences in terms of T-cell metabolism, we performed metabolic analysis using a seahorse flux analyzer. After repeated Ag-stim, 4-1BB and 79A40CAR showed a predominant oxidative phosphorylation metabolic pathway (Fig1A) and higher spare respiratory capacity (SRC), which has been correlated with mitochondrial functionality (Fig1B), than CD28CAR. After repeated Ag-stim, 4-1BB and 79A40CAR demonstrated higher ECAR than CD28CAR.
Because we observed differences in the expression of T-cell differentiation phenotype, we performed cell sorting to evaluate T-cell metabolism of each differentiation phenotype. After applying repeated Ag-stim to T CM and T EM-derived CD19CAR-T, mitochondrial functionality was evaluated using Mitotracker. T CM- (Fig1C) and T EM- (Fig1D) derived 4-1BB and 79A40CAR had more functional mitochondria and fewer dysfunctional mitochondria compared to CD28CAR.
Conclusion: 79A40CAR seems to preserve the mitochondrial oxidative phosphorylation-dominant metabolic pathway and supply energy demands for a longer period, especially when compared to CD28CAR. 79A40CAR showed functionally very similar to 4-1BBCAR. We showed that CD19CAR-Ts had different metabolic pathway according to the different co-stimulatory domains. To reveal additional metabolic pathway differences between 4-1BBCAR and 79A40CAR, we are now conducting proteomics analysis.
Disclosures
Kiyoi:Asahi Kasei: Research Funding; SymBio: Honoraria; AstraZeneca: Honoraria; Ono Pharmaceuticals: Honoraria; Eisai: Honoraria, Research Funding; Sumitomo Dainippon Pharma: Research Funding; Amgen: Honoraria; Nippon Kayaku: Honoraria; Zenyaku Kogyo: Research Funding; Bristol-Myers Squibb: Honoraria; Novartis: Honoraria; Chugai: Honoraria, Research Funding; Astellas Pharma: Honoraria, Research Funding; CURED: Research Funding; AbbVie: Honoraria, Research Funding; Daiichi Sankyo: Honoraria, Research Funding; Perseus Proteomics: Research Funding; Meiji Seika: Honoraria; Pfizer: Honoraria; Otsuka Pharmaceutical: Research Funding; Kyowa Hakko Kirin: Research Funding; Towa: Honoraria; MSD: Honoraria; Pharma Essentia Japan: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal